Baking Soda and Vinegar Fireworks
It’s time for some celebratory fireworks! With this baking soda and vinegar experiment we bring the excitement of the fireworks into our lessons with an exciting chemical reaction that kids of all ages will enjoy. We’ve included simple lessons, plus extensions that get into some fascinating chemistry (with a bit of math) to learn about measuring chemical reactions.
Summer Science – Measuring a Chemical Reaction

Disclaimer: This article may contain commission or affiliate links. As an Amazon Influencer I earn from qualifying purchases.
Not seeing our videos? Turn off any adblockers to ensure our video feed can be seen. Or visit our YouTube channel to see if the video has been uploaded there. We are slowly uploading our archives. Thanks!
Watching the fireworks is a popular celebratory activity through many cultures as a highlight of many festivals and celebrations. Today we are capturing that magic with a little baking soda and vinegar experiment.
Easily turn this into an experiment to celebrate any event by adjusting the colours of your glitter and mold, or just use a rainbow of colours and celebrate just because science is awesome!
Video of Fireworks Experiment
Check out the video of this fireworks chemistry experiment. If you can’t see the video, please turn off your adblockers as they also block our video feed. You can also check the STEAM Powered Family YouTube channel to see our videos.
Baking Soda and Vinegar
This is far from the first time we have done an experiment with sodium bicarbonate (aka baking soda) and vinegar. The bubbly, erupting reaction is easy, budget friendly, and so much fun.
Learn more!
Check out more baking soda and vinegar reactions here
The Science
Whenever we do any of these activities I love to talk about the science and reaction as we do it. It always amazes me the incredible, curious minds of kids, and the thoughtful and interesting questions they ask as we do science together.
This activity explores the popular baking soda and vinegar reaction, which is a simple acid-base chemical reaction. Vinegar or Acetic Acid has the chemical formula CH3COOH . Baking soda is a base also known as Sodium Bicarbonate and has the chemical formula NaHCO3 . During this reaction the products are sodium acetate ( C2H3NaO2 ). Sodium acetate is made of 1 sodium ion, 2 carbon atoms, 3 hydrogen atoms, and 2 oxygen atoms. The other products are water ( H2O) and carbon dioxide ( CO2 ). Carbon dioxide is the gas that causes the bubbling during the reaction.
Baking Soda and Vinegar Chemical Formula
C2H4O2 + NaHCO3 -> NaC2H3O2 + H2O + CO2
vinegar + sodium bicarbonate -> sodium acetate + water + carbon dioxide
What is an Element?
To dig in and learn more about elements, explore this comprehensive resource: What is an Element?
Supplies
For this experiment you will need:
Baking soda (sodium bicarbonate)
Vinegar
Star cookie cutters
Red, Blue and Silver glitter*
Spoon
Small Squeeze bottle, syringe or pipette (for vinegar)
Tray or large dish
Plastic covering for the table (optional)
*A NOTE ON GLITTER – Glitter has become very controversial due to the negative impacts on the environment, but I know many of you still love a little sparkle in your projects! When it comes to glitter, I believe it is always optional. None of my projects require it. But if you decide you do want a little sparkle and shine in your creations, I highly recommend using a biodegradeable glitter. There are lots of fantastic options available.
Depending on the age and ability of your children you may also want to provide them with protective equipment such as safety glasses (no one wants vinegar in their eye!) or gloves (vinegar in hang nails or little cuts can hurt).
Always do these experiments under responsible adult supervision.
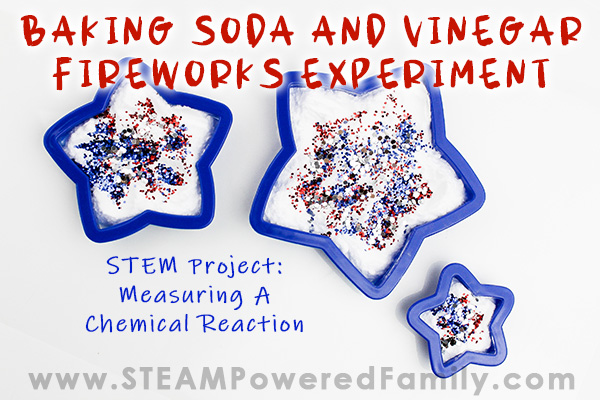
Directions
If needed, place a table cloth or plastic covering on the table to protect it and put on any safety equipment.
Fill your squeeze bottle with vinegar, or prepare a small bowl with vinegar if you are using syringe or pipette. Set to the side.
Place the cookie cutters in the tray or large dish.
Add about one heaping tablespoon of baking soda into the cookie cutter. Flatten it a bit with the spoon.
Sprinkle the top of the baking soda with your sprinkles. Or for a fun surprise, add the sprinkles, then add a bit of baking soda to the top of the sprinkles to hide them. This way they will be revealed by the reaction.
Now carefully add a few drops of vinegar. Start slow so you can really see the reaction. Add more vinegar as needed to continue the reaction.
The glitter gives it a really cool fireworks, sparkly effect!
Listen carefully, what do you hear? You can hear the reaction in our video too. What do you think is making that noise?

Pro Tip! Add a bit of dish soap to the inside of the cookie cutter with the baking soda. Adding this extra variable, the soap, will really add to the bubbles and eruption. Ask your kids why they think this happens.
Fireworks Printable
Add some fun to your activity with this Fireworks printable, free for STEAM Powered Family mailing list subscribers!
Math Extension Activity
Now we have done the basic experiment, let’s ramp up the learning a bit with an extension idea that will put our young scientists critical thinking and deductive reasoning skills to work.
Measuring the Chemical Reaction
Set your star cookie cutter in a small bowl or dish you can fit on a kitchen scale. Weigh the dish and cookie cutter together and note the weight. Now add in one tablespoon of baking soda and record the total, final weight. You will need this number in a moment.
Now place a small cup on the scale. Note the weight of the cup.
Add 50 mL of vinegar to the glass. Note the new weight. Now calculate the weight of the vinegar without the cup the same way we calculated the weight of the baking soda in the bonus activity. Record this number.
Time for math! Now add the weight of the vinegar without the cup to the total weight of the dish with cookie cutter and baking soda. That is the total starting mass of your chemical reaction. Record this number!
Start the reaction by pouring the vinegar onto the baking soda and watch the reaction.
Once the reaction is completely finished, and you don’t see any bubbles anymore, place the dish on the scale and take a final measurement.
Compare the starting mass of the chemical reaction with the final mass. How did the numbers change? What do you think caused this change in mass? Where did the missing mass go?
Ready to dive into more fun acid-base reactions? Including some really delicious ones? Check out these activities!











